Chromosome Segregation and Kinetochore research
Highlights
Activities
During mitosis, kinetochores orchestrate chromosome transmission from the mother into the daughter cells. Kinetochores are protein complexes containing 60 (yeast) to >100 components (humans). At mitotic entry, kinetochores assemble on the centromeres (CEN) of the replicated chromosomes (sister chromatids) to bi-orient them on the mitotic spindle, a dynamic array of centrosomes (spindle poles), microtubules and microtubule-associated proteins. Sister chromatid alignment is supervised by the spindle assembly checkpoint (SAC), which will delay cells in mitosis even if only one sister chromatid pair is not bi-oriented. Following satisfaction of the SAC and separation of all bi-oriented sisters (by dissolution of the cohesion linkages), kinetochores, motor proteins and spindle regression move the chromosomes into the daughter cells, generating offspring with a correct number of chromosomes. Errors made during chromosome segregation e.g., due to a lack or overexpression of a single kinetochore protein, can lead to daughter cells with an abnormal number of chromosomes (aneuploidy). Most often, aneuploidy results in death (e.g., the termination of a developing fetus). However, depending on which chromosome is affected, aneuploid organisms can be viable but will suffer from physical and mental disabilities, as exemplified by the Down syndrome. As most solid tumors are aneuploid it has been hypothesized that chromosome missegregation drives or supports the cancer transformation process. By identifying, functionally dissecting, and structurally analyzing kinetochore proteins and their regulators we can better understand the basis of aneuploidy disease, including cancer, and convert major players into cancer biomarkers and anticancer drug targets.<br><br> As chromosome segregation and the proteins involved are evolutionary conserved we study them in the yeast <i>Saccharomyces cerevisiae.</i> Using yeast allows for a quick genetic identification of proteins involved in chromosome segregation and for dissecting functional relationships between the involved proteins. In addition, progression through the yeast cell cycle can easily be manipulated with small compounds or via mutations in regulatory proteins. Importantly, insights obtained with yeast can be extrapolated to human cells.<br><br> Besides kinetochores we also study proteins regulating rDNA segregation in <i>Saccharomyces cerevisiae</i>. At anaphase entry, kinetochores initiate the segregation of >99% of the genome. <br>However, the rDNA locus separates at the end of anaphase. This is because cells must actively transcribe rDNA to generate ribosomes and sustain viability. In anaphase, rDNA becomes repressed allowing for local condensation and segregation before the next cell cycle initiates. Hence, the segregation of the genome is coordinated in time and space.<br><br> To study chromosome segregation we implement a multi-dimensional approach. To identify new proteins involved in chromosome segregation we perform protein affinity purifications and yeast two-hybrid screens. Alternatively, we identify new proteins and establish functional relationships with established kinetochore proteins using robotic genetic interaction screens (SGA). Protein localization and recruitment studies are performed by ChIP (Chromatin Immunoprecipiation), ChIP-CHIP (ChIP followed by microarray (CHIP) hybridization) and quantitative live-cell fluorescence imaging. <br>4D based protein localization, activity and copy number analyses during cell division are performed using time-lapse widefield DeltaVision deconvolution fluorescence microscopy or spinning disk confocal microscopy. Protein turnover in yeast is probed by FRAP (Fluorescence Recovery after Photobleaching). Phenotypic effects of mutations on chromosome segregation and mitosis are studied in synchronous cell cycles by indirect immunofluorescence microscopy, chromosome spreads, study of sister chromatid alignment using integrated pericentromeric GFP arrays, and FACS analysis (to probe the DNA status of the cells). At the molecular level, we study yeast protein activity biochemically in yeast cell extracts or in <i>in vitro</i> assays using recombinant proteins produced in bacteria, yeast or insect cells. Assays include co-IPs, <i>in vitro</i> protein-centromere binding studies using gel retardation analysis, <i>in vitro</i> protein reconstitution and affinity measurements using ITC or chromatography, kinase assays followed by mass spectroscopic identification of phosphorylated residues. Structural protein studies are performed by hydrodynamics (gel filtration plus glycerol gradient density velocity ultracentrifugation) and crystallographic analysis of recombinant proteins following crystallization at our in-house crystallography facility.<br><br> By integrating a variety of methodologies we wish to obtain a most detailed understanding of the proteins and regulators that drive chromosome segregation in yeast, and ultimately, in human cells.
-
Research projects
Research projects
1) Activity and regulation of Cnn1 at yeast kinetochores
Cnn1 is a conserved kinetochore protein (CENP-T in humans) that localizes to centromeric chromatin via its C-terminal histone fold domain (Figure 2). Its N-terminal tail extends inside the kinetochore and binds to the globular heads of the Spc24-Spc25 dimer of the Ndc80 complex (further comprises Ndc80 and Nuf2). The binding of Cnn1 to Spc24-Spc25 is regulated by phosphorylation (Cdc28 and Mps1 kinases) and stabilizes the Ndc80 complex at the kinetochore. However, to bind to the Ndc80 complex, the tail of Cnn1 must compete with the tail of Dsn1, a subunit of the tetrameric Mtw1 complex (Mis12 complex in humans). From G1 to metaphase, the high copy number of the Mtw1 complex and phospho-inhibition of the Cnn1 tail favors Mtw1-Ndc80 complex binding, creating a rigid kinetochore structure. At the metaphase-anaphase transition, Cnn1 levels at kinetochores increase equaling those of the Mtw1 and Ndc80 complexes. In addition, the degradation of Mps1, decrease in Cdc28 activity due to cyclin degradation, and an increased dephosphorylation of Cnn1 allow the latter to bind to the Ndc80 complex, resulting in a looser kinetochore structure. The latter may promote the binding of the Ndc80 complex to microtubules and to the microtubule-surrounding Dam1 ring complex, thereby supporting the transduction of forces generated by microtubule depolymerization that are required to mediate chromosome segregation.
We study at the molecular level how Cnn1 activity and levels at kinetochores are controlled during the cell cycle. This is important as CENP-T is misexpressed in myriad tumors. Using yeast will help to indicate how Cnn1/CENP-T anomalies can lead to aneuploidy, a hallmark of tumors.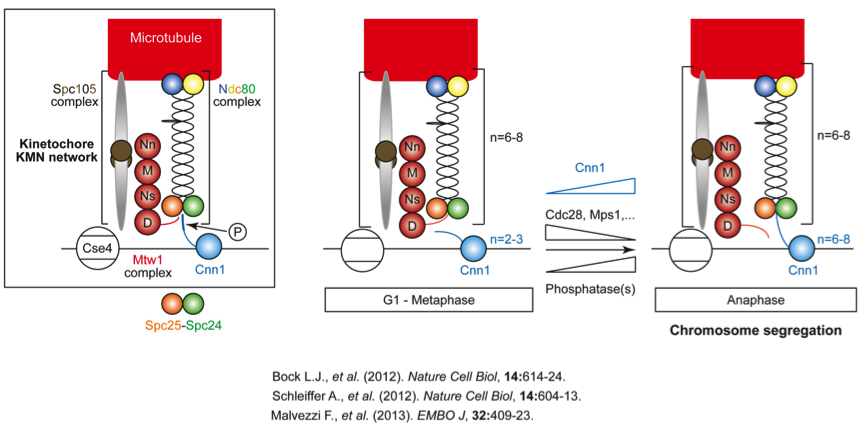
2) Regulation of rDNA segregation in yeast by the conserved Rio1 kinase
We recently localized the minimally studied yeast Rio1 kinase (RioK1-3 in humans) at the rDNA locus (also known as the nucleolus). We found that its nucleolar localization is dynamic and cell cycle stage dependent. Its enrichment at rDNA at anaphase onset indicated a role for Rio1 in rDNA segregation. Indeed, using a rio1 mutant we showed that yeast lacking nuclear Rio1 activity is unable to segregate the rDNA locus and finalize genome transmission at exit from mitosis. We found that the Rio1 kinase, in parallel to the Cdc14 phosphatase, represses PolI-mediated rDNA transcription allowing for local recruitment of condensin and initiation of rDNA segregation (Figure 3). Our study of how Rio1 orchestrates chromosome segregation in time and space we will generate new insights into a most basic mitotic process and suggest how an anomalous expression of Rio1, as observed in various tumors, may underlie aneuploidy in dividing cells.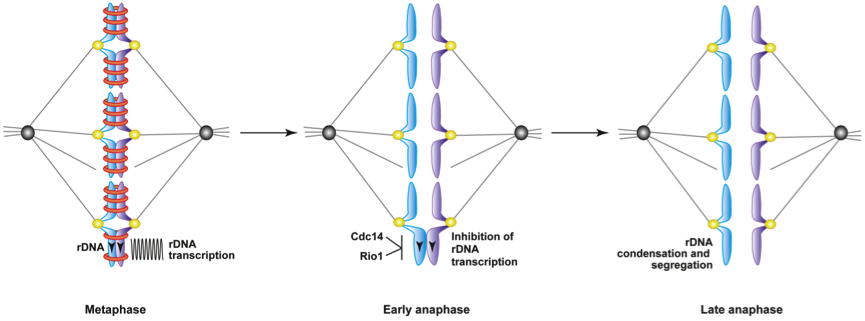
-
Publications
- Bock L.J., Pagliuca C., Kobayashi N., Grove R.A., Oku Y., Alfieri C., Golfieri C., Oldani A., Dal Maschio M., Bermejo R., Hazbun T.R., Tanaka T.U., De Wulf P. (2012). Cnn1 inhibits the interactions between the KMN complexes of the yeast kinetochore. Nature Cell Biology, 14:614-624.
- Nguyen T.L., Cera M.T., Pinto A., Lo Presti L., Hamel E., Conti P., Gussio R., De Wulf P. (2012). Evading Pgp activity in drug-resistant cancer cells: a structural and functional study of antitubulin furan metotica compounds. Molecular Cancer Therapeutics, 11:1103-1111.
- Cho-U-S, Corbett K.D., Al-Bassam J., Belizzi J.J.IIIrd, De Wulf P., Espelin C.W., Miranda J.J., Simons K., Sorger P.K., Harrison S.C. (2011). Molecular structures and interactions in the yeast kinetochore. Cold Spring Harbor Symposium in Quantitative Biology, 75:395-401.
- De Wulf P., Cheeseman IM (2010). Tension at EMBO's Aneuploidy Workshop. EMBO Reports, 11:727-729.
- Screpanti E., Santaguida S., Nguyen T.L., Silvestri R., Gussio R., Musacchio A., Hamel E., De Wulf P. (2010). A screen for kinetochore-microtubule interaction inhibitors identifies novel antitubulin compounds. PLoS ONE, 5:e11603.
-
Funding
- Italian Association for Cancer Research (AIRC).
- Italian Ministry of Health.
Past:- Italian Association for Cancer Research (AIRC)
- International Association for Cancer Research (AICR)
- European Molecular Biology Organization (EMBO)
- Fondazione Italiana per la Ricerca sul Cancro (FIRC)
- Italian Ministry of Health.
- European Institute of Oncology
- Development Therapeutics Program NCI/NIH
-
Staff

Chromosome Segregation and Kinetochore research UNIT
Director
Peter DE WULF

















